
BLOG
KATEGORİDEKİ DİĞER YAZILAR
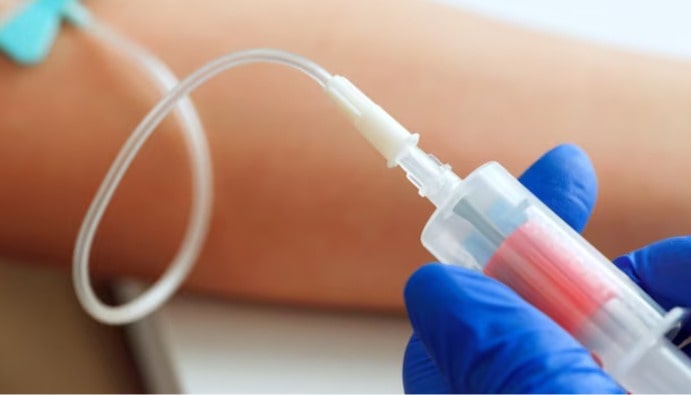
Materials used in blood-contact devices, such as intravenous catheters, hemodialysis sets, blood transfusion sets, vascular prostheses, need to be assessed for blood compatibility to ensure their safety. All materials are incompatible with blood to some degree as they can disrupt blood cells (hemolysis) or activate coagulation pathways (thrombogenicity) and/or the complement system.
Hemolysis is the process by which red blood cells (erythrocytes) break down and free hemoglobin is released into the blood. When a device that is incompatible with blood is used, the surface or material of this device can cause damage to cells when it interacts with blood cells. This is known as the hemolytic effect. The hemolytic effect can directly affect the biocompatibility of medical devices and can lead to serious health problems.
Hemolysis testing is a recommended test method for all devices or device materials except those in contact with intact skin or mucous membranes. The Hemolytic Effect (Blood Compatibility) (ISO 10993-4) test is performed to measure the damage to red blood cells when exposed to the material or extracts and compared to positive/negative controls.
Coagulation tests measure the effect of the sample on human blood clotting time. Hemolytic Effect (Blood Compatibility) Testing is recommended for all blood-contact devices.
The most common test for thrombogenicity is in vivo methods, and for medical devices that are not suitable for this test method, testing is required in each of the four categories covered by the ISO 10993-4 standard: coagulation, platelets, hematology and complement system.
For implant devices in contact with blood, complement activation testing is recommended. This in vitro test complements activation in human plasma as a result of exposure of plasma to the test substance or extract and aims to demonstrate that the test substance is capable of inducing a complement-induced inflammatoryimmune response in humans.
ISO 10993-4 sets certain limits on the interpretation of results from the hemolytic effect test. Generally, the hemolysis rate is evaluated as follows:
Nanolab Laboratories Group continues to provide services within the scope of Medical Device Analysis.
Contact us for more information.
You can follow us on LinkedIn for up-to-date news and posts about our services.
Follow our Instagram account to be informed about our latest blog posts.